-
Experimental evidence of librational vibrations determining the stability of calcium borohydride
A. Borgschulte, R. Gremaud, A. Züttel, P. Martelli, A. Remhof, A.J. Ramirez-Cuesta, K. Refson, E.G. Bardaji, W. Lohstroh, M. Fichtner, H. Hagemann and M. Ernst
Physical Review B, 83 (2) (2011), p24102


DOI:10.1103/PhysRevB.83.024102 | unige:14989 | Abstract | Article PDF
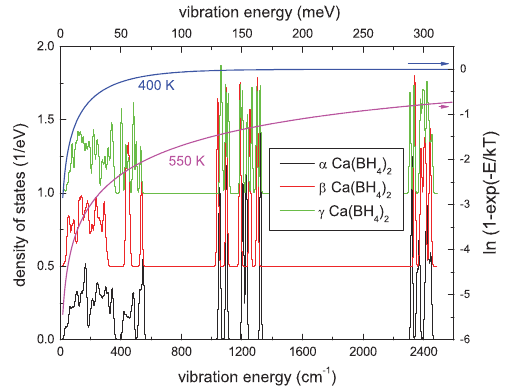
The high energy of hydrogen vibrations in solids is the origin of their strong impact on thermodynamic properties. The peculiar structure of complex hydrides amplifies this impact. We shed light on the vibrational properties of three allotropes of Ca(BH4)2 using density-functional theory calculations, infrared spectroscopy, and inelastic neutron scattering. We show that the vibrational properties of Ca(BH4)2 depend on the specific phase and are hitherto the origin of their differences in stability.
